Recent statistics indicate that approximately 70% of small molecules under development belong to Biopharmaceutics Classification System (BCS) II or IV poorly water-soluble drugs.
At Crystal Pharmatech, we utilize advanced amorphous solid dispersion (ASD) technology, including spray drying (SD) and hot melt extrusion (HME) to help our clients to solve the following challenges:
Low bioavailability
Long pharmacokinetic onset time
High inter- and intro-patient variability of plasma concentration
Food effect
Dose disproportionality
Complicated formulations and processes
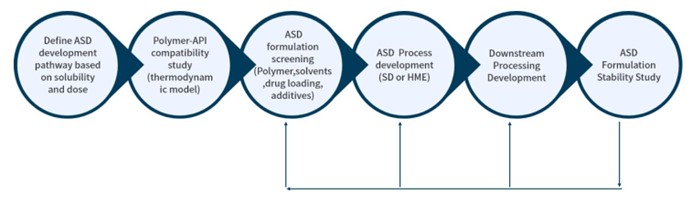
Spray Drying
The spray drying process is a particle engineering technique that is a proven approach to improving the solubility of poorly soluble compounds. It has been widely used to enhance bioavailability in drug manufacturing for the following advantages:
Scalable and cost-effective technique
Rapid, continuous process
Consistent particle size control
Ability to handle temperature-sensitive molecule
Long-term stability
Even spray drying is an effective way for solving bioavailability challenges, but it requires strong expertise in process design and development. The core technical team at Crystal Pharmatech has solid knowledge and expertise in the fundamentals and scaling-up principles for spray drying process and commercialized several amorphous solid dispersion solid dosage form drug products. With our deep understanding of all quality and regulatory requirements and world-class manufacturing and analytical equipment, we are confident that we can support amorphous solid dispersion formulation development world widely at high speed and with high quality.
Hot Melt Extrusion
HME is widely recognized as an effective technology that enhances the bioavailability of poorly soluble APIs, ultimately improving patient compliance and making the manufacturing process more efficient.
Increase development success rate of poorly soluble drugs
Optimize product efficacy, safety, and release properties
Well suitable for thermolabile products, as processing time is only minutes
Low dust formation (advantage over spray drying), safe handling of highly potent drugs
Solvent-free process (advantage over spray drying)
Relatively high drug load possible
Predictable and easy up-scale
Crystal Pharmatech's technical team has a solid background in polymer materials, chemical engineering, and mechanical engineering, a deep understanding of the principles of rheology and engineering, and industrial, advanced expertise in hot melt extrusion technology. We have successful experiences developing immediate and controlled release formulations and processes, scaling them up and commercializing them. Like spray drying, we have world-class bench-to-production equipment and analytical instruments for hot melt extrusion. We hope to help innovative drug hunters overcome the challenges on poorly water-soluble drugs with hot melt extrusion technology.