Published Mar 12, 2026
We want to move fast to first-in-human. How quickly can formulation and clinical supply be ready for IND?

In the high-stakes world of drug development, speed is the ultimate currency. But as the saying goes: "If you want to go fast, go alone; if you want to go far, go with a solid CMC strategy."
For a conventional oral solid dosage project, our standard timeline from the start of formulation development to the release of the CTM batch is typically 26 weeks.
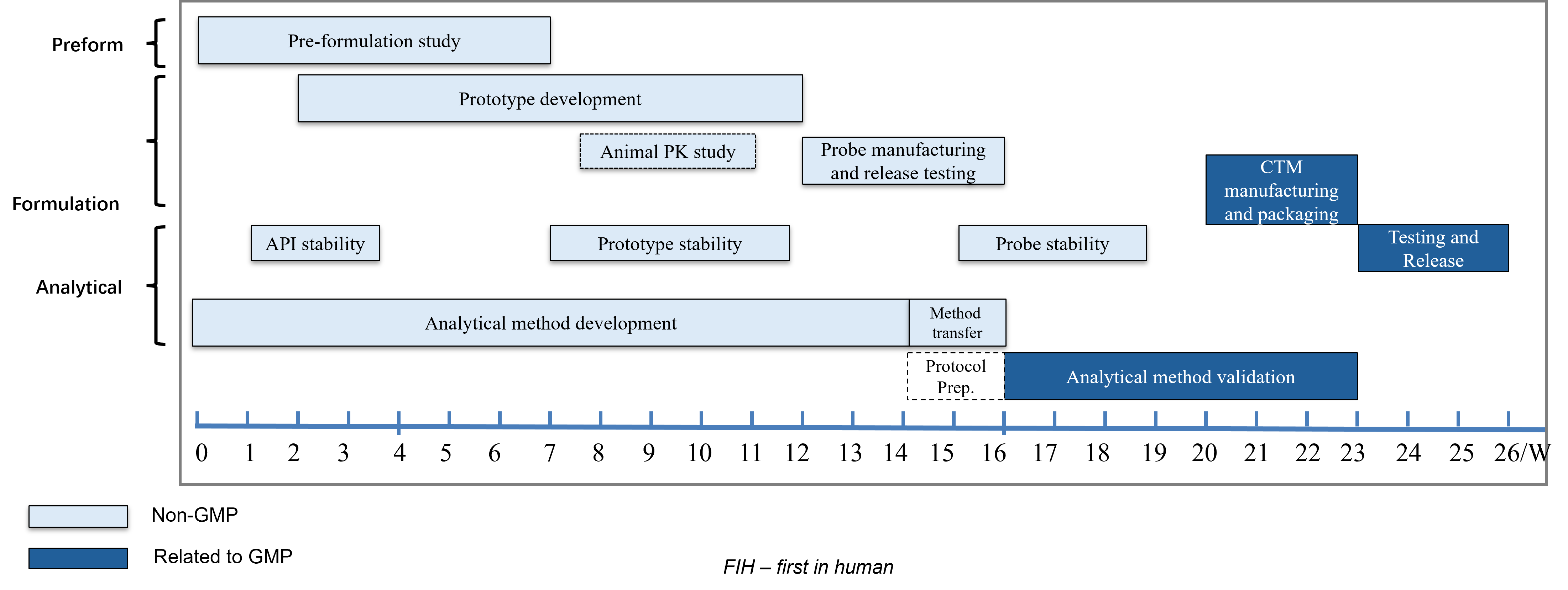
Weeks 1-7:
The Science: This stage is dedicated to physicochemical characterization (solubility, pKa, polymorphism) and pre-formulation stability profiling.
Strategic Intent: We identify potential "red flags" in the API early. By starting Analytical Method Development on Day 1, we ensure that every subsequent data point is supported by a robust, phase-appropriate method.
Weeks 8-16:
The Science: We move from theoretical design to Prototype Development and Animal PK studies. This creates an essential feedback loop to ensure in-vivo performance matches our target product profile (TPP).
Risk Reduction: The production of a Probe Batch (non-GMP) acts as a technical rehearsal for the GMP campaign, flushing out scale-up challenges before they become costly delays.
Weeks 16-23
The Science: This is the pivot from "Research" to "Compliance." We execute Analytical Method Validation in parallel with CTM (Clinical Trial Material) Manufacturing.
Compliance: Strict adherence to GMP standards during manufacturing and packaging ensures that the drug substance is transformed into a high-quality drug product ready for human dosing.
Weeks 23-26
The Science: The final phase involves comprehensive Testing and Release, including stability testing of the Probe and CTM batches.
Final Release: A rigorous QA review culminates in the release of the Certificate of Analysis (CoA), marking the final green light for FIH initiation.
For projects with specific requirements, further discussions are needed to assess the feasibility of shortening the timeline.
Remember that speed also include "First-Time-Right" formulation decision. Wrong decisions will cost much more delays for going back and forth.
Every molecule is unique. If your API has poor solubility (BCS Class II/IV) or is chemically unstable, the "fastest" path is often the one that invests an extra 4 weeks upfront to avoid a 12-month delay caused by a failed stability study later.
Found this blog helpful? Whether you have a specific technical question or need expert support for your current pipeline, our team is here to help. Complete the form, and we'll reach out to discuss how our expertise can drive your success.
Our expertise spans three specialized platforms:
Small Molecule
Crystal Bio Solutions
Crystal NAX
By providing your e-mail address, you agree to receive an e-mail response from Crystal Pharmatech to your inquiry. The information you submit will be governed by our Privacy Policy.
Subscribe to be the first to get the updates!