News
Crystal Pharmatech | Reflecting on 2025
21 Feb 2026
 ELISpot Assays
Vaccine efficacy and characterization of cell-mediated immune response
ELISpot Assays
Vaccine efficacy and characterization of cell-mediated immune response
 Solid-State NMR Analysis
Nuclear Magnetic Resonance (NMR) Spectroscopy is a ubiquitous analytical technique utilized across a wide range of disciplines.
Solid-State NMR Analysis
Nuclear Magnetic Resonance (NMR) Spectroscopy is a ubiquitous analytical technique utilized across a wide range of disciplines.
 Animal Dosing Vehicle Selection
Choosing an optimal vehicle for animal PK and Tox studies requires multidisciplinary skillsets and a sound understanding of the physicochemical properties of your compound. Controlling solid and solut...
Animal Dosing Vehicle Selection
Choosing an optimal vehicle for animal PK and Tox studies requires multidisciplinary skillsets and a sound understanding of the physicochemical properties of your compound. Controlling solid and solut...
 Analytical Research and Quality Control
We will provide our valued customers with analytical chemistry research and quality control services for NCEs throughout their life cycle from the early clinical stage to commercial manufacturing. Ana...
Analytical Research and Quality Control
We will provide our valued customers with analytical chemistry research and quality control services for NCEs throughout their life cycle from the early clinical stage to commercial manufacturing. Ana...
 Molecular Biology
Cell/gene therapy, RNA vaccines, gene editing therapy and biomarkers utilizing qPCR, ddPCR, RNA-Seq, NanoString, and NGS
Molecular Biology
Cell/gene therapy, RNA vaccines, gene editing therapy and biomarkers utilizing qPCR, ddPCR, RNA-Seq, NanoString, and NGS
 Clinical and Commercial Manufacturing
We provide flexible and reliable OSD manufacturing services using versatile technologies to address your formulation challenges.
Clinical and Commercial Manufacturing
We provide flexible and reliable OSD manufacturing services using versatile technologies to address your formulation challenges.
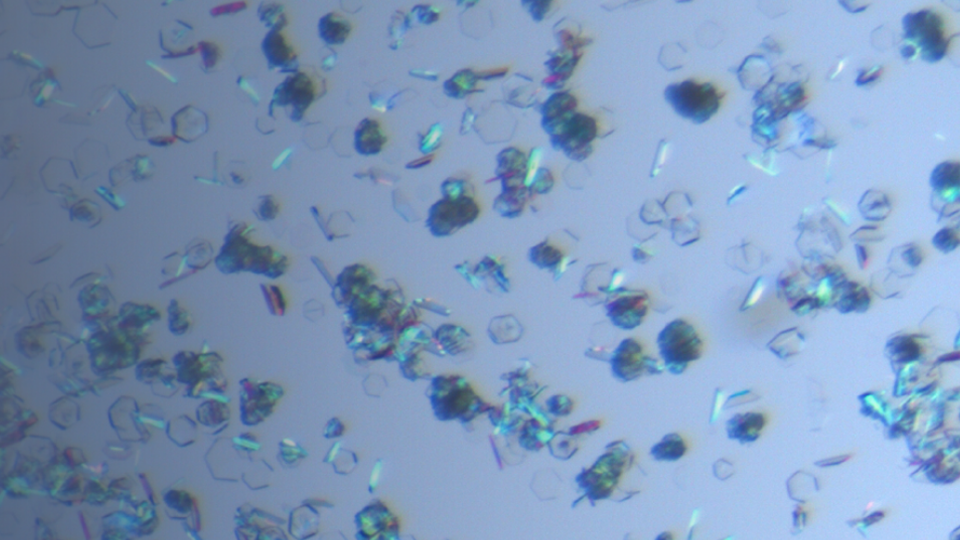 Amorphous Solid Dispersions
Amorphous solid dispersions have been shown to alter the properties of an API, including solubility, dissolution, and bioavailability. A variety of polymers or additives are available, and screening i...
Amorphous Solid Dispersions
Amorphous solid dispersions have been shown to alter the properties of an API, including solubility, dissolution, and bioavailability. A variety of polymers or additives are available, and screening i...
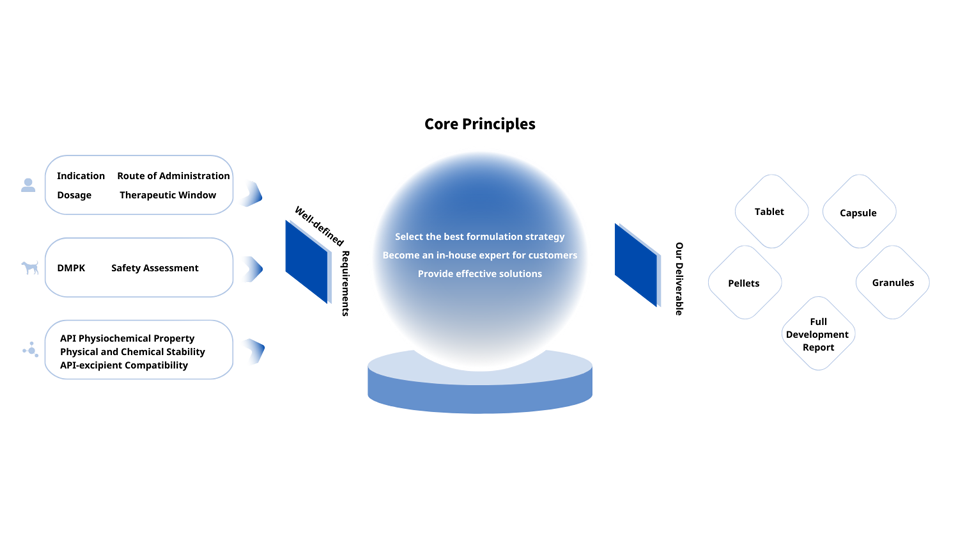 First-Time-Right Formulation Development
We guide our customers into making smarter formulation choices. Ultimately, this reduces the risk of clinical failure and ensures your product reaches the clinic rapidly via the most efficient route.
First-Time-Right Formulation Development
We guide our customers into making smarter formulation choices. Ultimately, this reduces the risk of clinical failure and ensures your product reaches the clinic rapidly via the most efficient route.
 Oral Solid Dosage Forms
Most small molecule drugs are formulated as oral solid dosage (OSD) forms, such as tablets or capsules. OSD has been an important part of the global pharmaceutical market. The formulation team of Crys...
Oral Solid Dosage Forms
Most small molecule drugs are formulated as oral solid dosage (OSD) forms, such as tablets or capsules. OSD has been an important part of the global pharmaceutical market. The formulation team of Crys...
 Clinical Supply
Crystal Pharmatech provides a one-stop Clinical Supply Service from pre-clinical to post-marketing. Services provided: Primary packagingcGMP environment; cGMP requirements; Blistering, bottling, capsulat...
Clinical Supply
Crystal Pharmatech provides a one-stop Clinical Supply Service from pre-clinical to post-marketing. Services provided: Primary packagingcGMP environment; cGMP requirements; Blistering, bottling, capsulat...
 Amorphous Solid Dispersion (Spray Drying & Hot-melt Extrusion)
Recent statistics indicate that approximately 70% of small molecules under development belong to Biopharmaceutics Classification System (BCS) II or IV poorly water-soluble drugs. At Crystal Pharmatech...
Amorphous Solid Dispersion (Spray Drying & Hot-melt Extrusion)
Recent statistics indicate that approximately 70% of small molecules under development belong to Biopharmaceutics Classification System (BCS) II or IV poorly water-soluble drugs. At Crystal Pharmatech...
 Pediatric Formulation (Mini-tablet)
The development of acceptable, palatable pediatric formulations is undergoing transformation within the industry today, driven by patient needs and regulatory requirements. Therefore, the demand for a...
Pediatric Formulation (Mini-tablet)
The development of acceptable, palatable pediatric formulations is undergoing transformation within the industry today, driven by patient needs and regulatory requirements. Therefore, the demand for a...
Subscribe to be the first to get the updates!