News
A Significant Step Toward Greater Clarity in Polymorph Control for Innovative Drugs
28 Jan 2026
 Developability Assessment
For lead compounds or candidate compounds, Crystal pharmatech can make an objective evaluation of the development potential and risks of drugs through comprehensive and accurate evaluation of the physicochemical properties of compounds, combined with a variety of mature development evaluation models and GastroPlus® software.
Developability Assessment
For lead compounds or candidate compounds, Crystal pharmatech can make an objective evaluation of the development potential and risks of drugs through comprehensive and accurate evaluation of the physicochemical properties of compounds, combined with a variety of mature development evaluation models and GastroPlus® software.
 CATUG and Crystal Bio Establish Strategic Partnership, Launching “CATUG-Crystal” Joint Lab Dedicated to Advanced Nucleic Acid Analytical Services
CAMBRIDGE, Massachusetts & CRANBURY, New Jersey, February 21, 2024, – CATUG Inc. (CATUG) and Crystal Bio, a member of Crystal Pharmatech, announced today a long-term strategic partnership to prov...
CATUG and Crystal Bio Establish Strategic Partnership, Launching “CATUG-Crystal” Joint Lab Dedicated to Advanced Nucleic Acid Analytical Services
CAMBRIDGE, Massachusetts & CRANBURY, New Jersey, February 21, 2024, – CATUG Inc. (CATUG) and Crystal Bio, a member of Crystal Pharmatech, announced today a long-term strategic partnership to prov...
 Applying PBPK and PBBM Across R&D: From Early Insights to Formulation
Applying PBPK and PBBM Across R&D: FromEarly Insights to FormulationDrug-delivery programs face recurring hurdles: frequent dosing, nighttimecoverage, and site-specific absorption. Controlled- and...
Applying PBPK and PBBM Across R&D: From Early Insights to Formulation
Applying PBPK and PBBM Across R&D: FromEarly Insights to FormulationDrug-delivery programs face recurring hurdles: frequent dosing, nighttimecoverage, and site-specific absorption. Controlled- and...
 Mol2Med™ Developability Assessment: Material-Sparing, Rapid Studies, and Developability Strategies
From early developability to late-stage specifications—polymorph/salt-cocrystal strategy, SCXRD/MicroED solutions, solvent & pathway design, drug-product form control, ASD crystalline-form limits...
Mol2Med™ Developability Assessment: Material-Sparing, Rapid Studies, and Developability Strategies
From early developability to late-stage specifications—polymorph/salt-cocrystal strategy, SCXRD/MicroED solutions, solvent & pathway design, drug-product form control, ASD crystalline-form limits...
 Meet Crystal Pharmatech at DCAT Week 2026
Crystal Pharmatech is returning to DCAT Week with expanded capabilities. We have grown into a global CDMO with 300+ scientists across the US, Canada, and China, having supported over 2,000 NCEs to dat...
Meet Crystal Pharmatech at DCAT Week 2026
Crystal Pharmatech is returning to DCAT Week with expanded capabilities. We have grown into a global CDMO with 300+ scientists across the US, Canada, and China, having supported over 2,000 NCEs to dat...
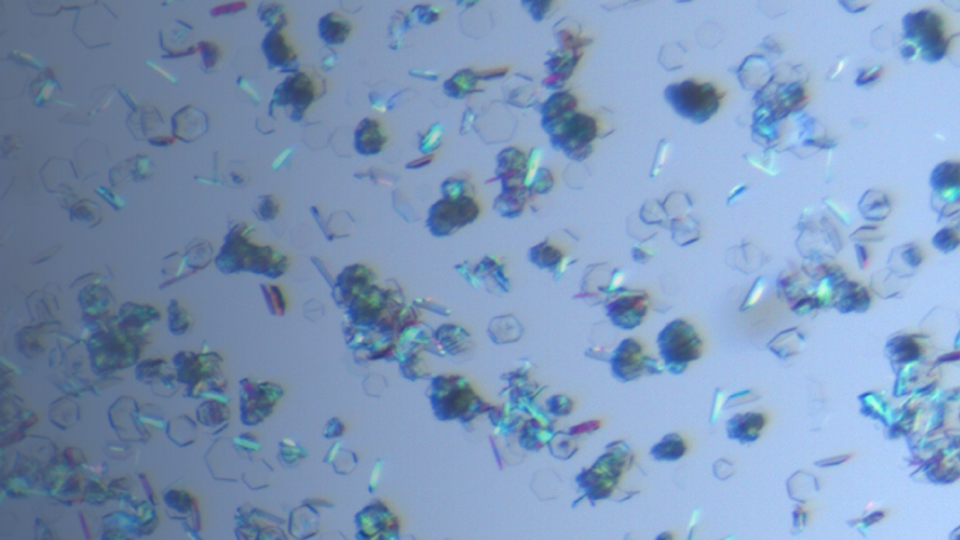 Single Crystal Growth & Structure Determination
Single crystal growth and structure determination technology are the main methods to determine the absolute configuration of drug molecules and identify the crystal forms absolutely. The single crysta...
Single Crystal Growth & Structure Determination
Single crystal growth and structure determination technology are the main methods to determine the absolute configuration of drug molecules and identify the crystal forms absolutely. The single crysta...
 Bioavailability Enhancement for Insoluble Compounds & PROTAC & Oral Peptides
With the ever-increasing demand for the absorbability of small-molecule oral innovative drugs, more and more new molecules have hydrophobic properties, resulting in very low solubility. According to the latest statistics, about 70% of the small-molecule drugs under research are insoluble substances of BCS II or IV.
Bioavailability Enhancement for Insoluble Compounds & PROTAC & Oral Peptides
With the ever-increasing demand for the absorbability of small-molecule oral innovative drugs, more and more new molecules have hydrophobic properties, resulting in very low solubility. According to the latest statistics, about 70% of the small-molecule drugs under research are insoluble substances of BCS II or IV.
 Events
Events
 Case Study 3: Atorvastatin - Crystalline Form Change In Late Development
Atorvastatin (CI-981) is an HMG CoA reductase inhibitor marketed as Lipitor® (Fig. 8). As a BCS II drug, it has exhibited poor solubility and high permeability (Wu and Benet 2005). The compound was o...
Case Study 3: Atorvastatin - Crystalline Form Change In Late Development
Atorvastatin (CI-981) is an HMG CoA reductase inhibitor marketed as Lipitor® (Fig. 8). As a BCS II drug, it has exhibited poor solubility and high permeability (Wu and Benet 2005). The compound was o...
Subscribe to be the first to get the updates!