News
Crystal Pharmatech | Reflecting on 2025
21 Feb 2026
 Industry Shift: FDA Goes Vegan?
Approaching Drug Development With FinesseIn 2012, drug development specialists started an initiative to create predictive Oral Bioavailability Tools (OrBiTo). This initiative recognized the need to mo...
Industry Shift: FDA Goes Vegan?
Approaching Drug Development With FinesseIn 2012, drug development specialists started an initiative to create predictive Oral Bioavailability Tools (OrBiTo). This initiative recognized the need to mo...
 Analytical Research and Quality Control
We will provide our valued customers with analytical chemistry research and quality control services for NCEs throughout their life cycle from the early clinical stage to commercial manufacturing. Ana...
Analytical Research and Quality Control
We will provide our valued customers with analytical chemistry research and quality control services for NCEs throughout their life cycle from the early clinical stage to commercial manufacturing. Ana...
 Development and Production Application Cases of Amorphous Solid Dispersion Formulations (I) - Hot Melt Extrusion
Hot-melt extrusion (HME) is recognized as an optimal and efficient technique for manufacturing amorphous solid dispersions, characterized by its continuous processing, increased production efficiency,...
Development and Production Application Cases of Amorphous Solid Dispersion Formulations (I) - Hot Melt Extrusion
Hot-melt extrusion (HME) is recognized as an optimal and efficient technique for manufacturing amorphous solid dispersions, characterized by its continuous processing, increased production efficiency,...
 Overcoming Bioavailability Barrier to Deliver Protein Degraders
Speaker: Dr. Chong-Hui Gu, Head of CMC and QA, Foghorn Therapeutics
Overcoming Bioavailability Barrier to Deliver Protein Degraders
Speaker: Dr. Chong-Hui Gu, Head of CMC and QA, Foghorn Therapeutics
 Discovery & Development US 2025
We're heading to San Diego October 2-3 and bringing a team ready to talk First-Time-Right CMC for small molecules—from solid form to formulation and IND.Date: October 2-3Location: Sheraton San Di...
Discovery & Development US 2025
We're heading to San Diego October 2-3 and bringing a team ready to talk First-Time-Right CMC for small molecules—from solid form to formulation and IND.Date: October 2-3Location: Sheraton San Di...
 Clinical and Commercial Manufacturing
We provide flexible and reliable OSD manufacturing services using versatile technologies to address your formulation challenges.
Clinical and Commercial Manufacturing
We provide flexible and reliable OSD manufacturing services using versatile technologies to address your formulation challenges.
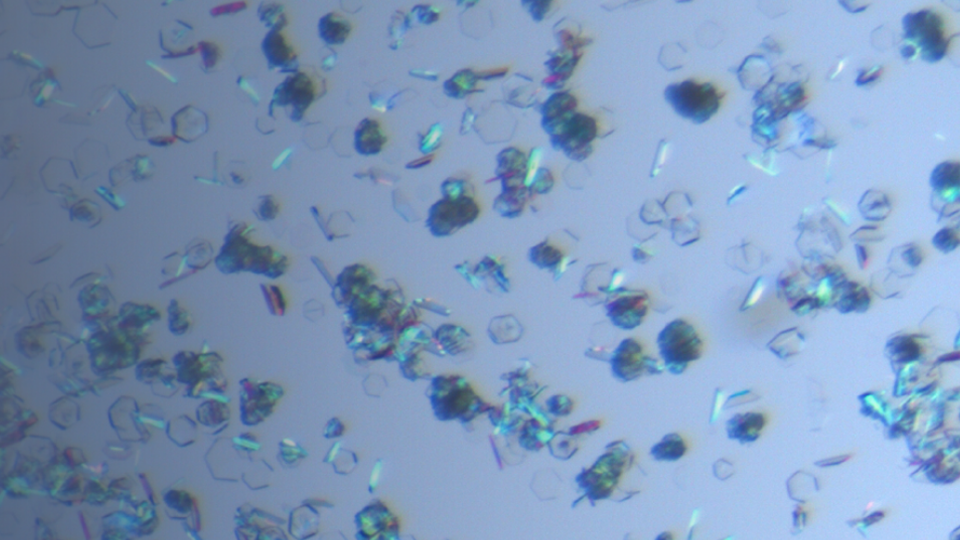 Amorphous Solid Dispersions
Amorphous solid dispersions have been shown to alter the properties of an API, including solubility, dissolution, and bioavailability. A variety of polymers or additives are available, and screening i...
Amorphous Solid Dispersions
Amorphous solid dispersions have been shown to alter the properties of an API, including solubility, dissolution, and bioavailability. A variety of polymers or additives are available, and screening i...
 Crystal Pharmatech's CDMO Business Unit - Crystal Formulation Services Received China Drug Product Manufacturing License, Achieving the Important Milestone in Formulation Capability
Crystal Formulation Services, a subsidiary of Crystal Pharmatech, has achieved an exciting new milestone in its formulation capabilities. The company was recently granted the Drug Manufacturing ...
Crystal Pharmatech's CDMO Business Unit - Crystal Formulation Services Received China Drug Product Manufacturing License, Achieving the Important Milestone in Formulation Capability
Crystal Formulation Services, a subsidiary of Crystal Pharmatech, has achieved an exciting new milestone in its formulation capabilities. The company was recently granted the Drug Manufacturing ...
 Formulation Development
Instead of one-size-fits-all platforms, Crystal Pharmtech applies a "First-Time-Right" strategy for formulation development to accelerate your molecule to First in Human (FIH) studies and onward to commercialization in a cost-effective and time-saving way.
Formulation Development
Instead of one-size-fits-all platforms, Crystal Pharmtech applies a "First-Time-Right" strategy for formulation development to accelerate your molecule to First in Human (FIH) studies and onward to commercialization in a cost-effective and time-saving way.
Subscribe to be the first to get the updates!