News
Crystal Pharmatech | Reflecting on 2025
21 Feb 2026
 Crystal Formulation Services' GMP Manufacturing Facility Successfully Passes the Remote Audit by US Client, Marking a Key Milestone for Its International Expansion
Today, Crystal Pharmatech announced that the GMP manufacturing facility of their CDMO business unit – Crystal Formulation Services (CFS) has successfully passed a remote audit conducted by a US clien...
Crystal Formulation Services' GMP Manufacturing Facility Successfully Passes the Remote Audit by US Client, Marking a Key Milestone for Its International Expansion
Today, Crystal Pharmatech announced that the GMP manufacturing facility of their CDMO business unit – Crystal Formulation Services (CFS) has successfully passed a remote audit conducted by a US clien...
 ASD Column | Mastering Stable Art: Unveiling Key Factors Influencing Physical Stability of Amorphous Solid Dispersions
/ Formulation Rightthe First Time /In recent years, amorphous solid dispersions (ASD) have attracted attention due to their ability to significantly improve the solubility of poorly soluble drugs. Sin...
ASD Column | Mastering Stable Art: Unveiling Key Factors Influencing Physical Stability of Amorphous Solid Dispersions
/ Formulation Rightthe First Time /In recent years, amorphous solid dispersions (ASD) have attracted attention due to their ability to significantly improve the solubility of poorly soluble drugs. Sin...
 Join Crystal Pharmatech for the Sino-American Pharmaceutical Professionals Association (SAPA) NE 26th Annual Conference
Join our team for the Sino-American Pharmaceutical Professionals Association (SAPA) NE 26th Annual Conference in Cambridge, MA on Saturday (6/8).
Join Crystal Pharmatech for the Sino-American Pharmaceutical Professionals Association (SAPA) NE 26th Annual Conference
Join our team for the Sino-American Pharmaceutical Professionals Association (SAPA) NE 26th Annual Conference in Cambridge, MA on Saturday (6/8).
 ASD Column | How to Select Polymers in Hot-melt Extrusion Process?
Hot melt extrusion (HME) technology is widely used in the development of amorphous solid dispersions (ASD) due to its advantages such as solvent-free processing, continuous manufacturing, high automat...
ASD Column | How to Select Polymers in Hot-melt Extrusion Process?
Hot melt extrusion (HME) technology is widely used in the development of amorphous solid dispersions (ASD) due to its advantages such as solvent-free processing, continuous manufacturing, high automat...
 Crystal Bio Welcomes Dr. Shiaw-Lin (Billy) Wu as Co-Founder and Chief Scientific Officer
Cranbury, New Jersey, January 17, 2024 - Crystal Bio, a key division of Crystal Pharmatech, is excited to announce Dr. Shiaw-Lin (Billy) Wu as its new co-founder and Chief Scientific Officer (CSO). Dr...
Crystal Bio Welcomes Dr. Shiaw-Lin (Billy) Wu as Co-Founder and Chief Scientific Officer
Cranbury, New Jersey, January 17, 2024 - Crystal Bio, a key division of Crystal Pharmatech, is excited to announce Dr. Shiaw-Lin (Billy) Wu as its new co-founder and Chief Scientific Officer (CSO). Dr...
 Crystal Pharmatech will be in San Diego from June 3-6 at BIO International Convention
Crystal Pharmatech senior leadership will be in San Diego from June 3-6 at BIO International Convention to discuss the latest in drug development.
Crystal Pharmatech will be in San Diego from June 3-6 at BIO International Convention
Crystal Pharmatech senior leadership will be in San Diego from June 3-6 at BIO International Convention to discuss the latest in drug development.
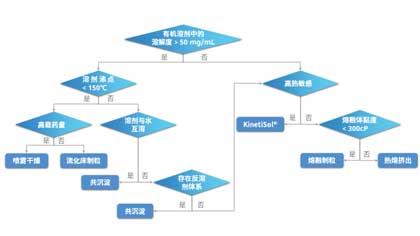 ASD Column | Understanding the ASD Preparation Methods and Selecting the Optimal Method for Solution
In the previous article Mastering the Art of Stability, we revealed the key factors influencing the physical stability of amorphous solid dispersions (ASDs) and recognized that choosing th...
ASD Column | Understanding the ASD Preparation Methods and Selecting the Optimal Method for Solution
In the previous article Mastering the Art of Stability, we revealed the key factors influencing the physical stability of amorphous solid dispersions (ASDs) and recognized that choosing th...
 Meet Crystal Pharmatech at Chinese Antibody Society 2024 Annual Conference
Join our team in Boston on Saturday for the Chinese Antibody Society 2024 Annual Conference.Catch up with Crystal Bio's CSO, Billy Wu, Ph.D., and our new CTO, Ye Gu, Ph.D. We will also be joined b...
Meet Crystal Pharmatech at Chinese Antibody Society 2024 Annual Conference
Join our team in Boston on Saturday for the Chinese Antibody Society 2024 Annual Conference.Catch up with Crystal Bio's CSO, Billy Wu, Ph.D., and our new CTO, Ye Gu, Ph.D. We will also be joined b...
 Crystal Bio Appoints Dr. Ye Gu as Co-founder, CTO, and Head of USA BD
Cranbury, NJ, May 14, 2024 – Crystal Bio, an innovative biologics analytical division of Crystal Pharmatech, is delighted to announce the appointment of Dr. Ye Gu as Co-founder, Chief Technology Officer, and Head of USA Business Development. Dr. Gu brings over 16 years of distinguished experience in biologics R&D and a robust track record of leadership in the biotechnology industry.
Crystal Bio Appoints Dr. Ye Gu as Co-founder, CTO, and Head of USA BD
Cranbury, NJ, May 14, 2024 – Crystal Bio, an innovative biologics analytical division of Crystal Pharmatech, is delighted to announce the appointment of Dr. Ye Gu as Co-founder, Chief Technology Officer, and Head of USA Business Development. Dr. Gu brings over 16 years of distinguished experience in biologics R&D and a robust track record of leadership in the biotechnology industry.
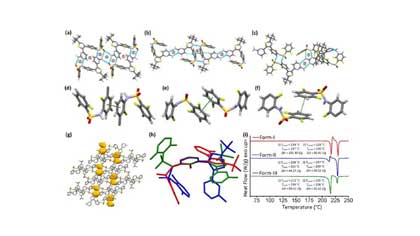 Polymorphs, Solvatomorphs and Hydrate of Dabrafenib
Explore the polymorphs, solvatomorphs, monohydrate, and perhydrate of anticancer drug dabrafenib (DBF). Detailed analysis from ACS study including crystal packing, DFT calculations, stability predictions, and implications for pharmaceutical solid-form screening at Crystal Pharmatech.
Polymorphs, Solvatomorphs and Hydrate of Dabrafenib
Explore the polymorphs, solvatomorphs, monohydrate, and perhydrate of anticancer drug dabrafenib (DBF). Detailed analysis from ACS study including crystal packing, DFT calculations, stability predictions, and implications for pharmaceutical solid-form screening at Crystal Pharmatech.
Subscribe to be the first to get the updates!