News
Crystal Pharmatech | Reflecting on 2025
21 Feb 2026
 Crystal Pharmatech's CDMO Business Unit - Crystal Formulations Services Successfully Passed EU QP Audit
Recently, Crystal Formulations Services (CFS), the CDMO Business Unit of Crystal Pharmatech has achieved a significant milestone by successfully passing the on-site audit conducted by the European Qua...
Crystal Pharmatech's CDMO Business Unit - Crystal Formulations Services Successfully Passed EU QP Audit
Recently, Crystal Formulations Services (CFS), the CDMO Business Unit of Crystal Pharmatech has achieved a significant milestone by successfully passing the on-site audit conducted by the European Qua...
 Meet Crystal Pharmatech at 15th Drug Discovery Strategic Summit (DDSS)
Accelerated development from discovery to the clinic. Catch up with Ridwan Islam, M. Pharm., Ph.D. in Boston from May 8-9 at the 15th Drug Discovery Strategic Summit.Date: May 8-9, 2024Location: Emba...
Meet Crystal Pharmatech at 15th Drug Discovery Strategic Summit (DDSS)
Accelerated development from discovery to the clinic. Catch up with Ridwan Islam, M. Pharm., Ph.D. in Boston from May 8-9 at the 15th Drug Discovery Strategic Summit.Date: May 8-9, 2024Location: Emba...
 Assessment of CQA in mRNA-LNP Modality
“Figure 1. mRNA encapsulated with LNP typically composed of encapture nucleic acids such as mRNA, siRNA, etc. as shown in the blue curve line by lipid nanoparticle, LNP as ionized, pegylated, phosph...
Assessment of CQA in mRNA-LNP Modality
“Figure 1. mRNA encapsulated with LNP typically composed of encapture nucleic acids such as mRNA, siRNA, etc. as shown in the blue curve line by lipid nanoparticle, LNP as ionized, pegylated, phosph...
 Shining Glory: Unveiling the “True Eye” behind the Veil of Drug Crystal Forms
Are you still troubled by the characterization of solid state?How to determine whether the prepared crystal form is the same?Compounds have polymorphism. Is it necessary to conduct impurity crystal fo...
Shining Glory: Unveiling the “True Eye” behind the Veil of Drug Crystal Forms
Are you still troubled by the characterization of solid state?How to determine whether the prepared crystal form is the same?Compounds have polymorphism. Is it necessary to conduct impurity crystal fo...
 Meet Crystal Pharmatech at Sorption Symposium North America 2024
Solid State Chemistry: Do You Like Solving Puzzles?Understanding the chemical composition and physical properties of crystalline API can be accomplished by using a variety of instruments and methods. ...
Meet Crystal Pharmatech at Sorption Symposium North America 2024
Solid State Chemistry: Do You Like Solving Puzzles?Understanding the chemical composition and physical properties of crystalline API can be accomplished by using a variety of instruments and methods. ...
 Crystal Pharmatech and Divamics Announce Strategic Collaboration to Accelerate Drug Development through AI, Crystal Form, and Formulation Innovation
Cranbury, New Jersey, USA – May 9, 2025 – Crystal Pharmatech Inc. Crystal Pharmatech and Divamics Inc. Divamics today announced a strategic collaboration to integrate AI-aided...
Crystal Pharmatech and Divamics Announce Strategic Collaboration to Accelerate Drug Development through AI, Crystal Form, and Formulation Innovation
Cranbury, New Jersey, USA – May 9, 2025 – Crystal Pharmatech Inc. Crystal Pharmatech and Divamics Inc. Divamics today announced a strategic collaboration to integrate AI-aided...
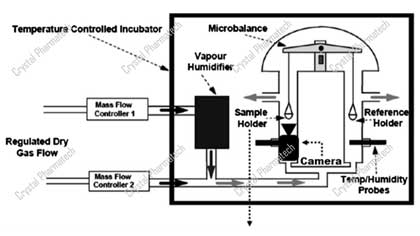 Applications of Dynamic Moisture Adsorption in Crystal Research
Content Overview1. Introduction2. Regulatory Requirements3. Application of Dynamic Vapor Sorption (DVS)3.1 Instrument Introduction3.2 Application 1 - Moisture Sorption Assessment3.3 Application 2 - Va...
Applications of Dynamic Moisture Adsorption in Crystal Research
Content Overview1. Introduction2. Regulatory Requirements3. Application of Dynamic Vapor Sorption (DVS)3.1 Instrument Introduction3.2 Application 1 - Moisture Sorption Assessment3.3 Application 2 - Va...
 CPHI North America - Meet Crystal Pharmatech at Booth 1441
Catch up with Crystal Pharmatech Subject Matter Expert Derik McCarthy in Philadelphia from May 7-9 at CPHI North America.We will be at Booth 1441 with LAVIANA PHARMA CO, LTD
CPHI North America - Meet Crystal Pharmatech at Booth 1441
Catch up with Crystal Pharmatech Subject Matter Expert Derik McCarthy in Philadelphia from May 7-9 at CPHI North America.We will be at Booth 1441 with LAVIANA PHARMA CO, LTD
 Parameter Sensitivity with GastroPlus
How can physiologically-based pharmacokinetic (PBPK) modeling maximize ADME performance and identify a path forward for GLP Tox and FIH formulations?GastroPlus incorporates in-vivo and in-vitro data t...
Parameter Sensitivity with GastroPlus
How can physiologically-based pharmacokinetic (PBPK) modeling maximize ADME performance and identify a path forward for GLP Tox and FIH formulations?GastroPlus incorporates in-vivo and in-vitro data t...
 Crystal Pharmatech Receives CNAS Reaccreditation, Scope Expansion, and Address Approval
Suzhou, China – August 11, 2025 – Crystal Pharmatech Co., Ltd. announced that its China Suzhou CRO business unit has successfully completed the re-assessment, scope expansion, and laboratory address...
Crystal Pharmatech Receives CNAS Reaccreditation, Scope Expansion, and Address Approval
Suzhou, China – August 11, 2025 – Crystal Pharmatech Co., Ltd. announced that its China Suzhou CRO business unit has successfully completed the re-assessment, scope expansion, and laboratory address...
 Application of Granularity Analysis in Crystal Typing Research
This article series is initiated and compiled by the Crystalline Cloud Solid-State Testing Center. Its aim is to create a platform for exchange and learning among industry peers. We intend to explore ...
Application of Granularity Analysis in Crystal Typing Research
This article series is initiated and compiled by the Crystalline Cloud Solid-State Testing Center. Its aim is to create a platform for exchange and learning among industry peers. We intend to explore ...
Subscribe to be the first to get the updates!