News
Crystal Pharmatech | Reflecting on 2025
21 Feb 2026
 Home
Crystal Pharmatech's Mol2Med™ First-Time-Right approach guarantees a robust API form and a scalable manufacturing process, culminating in a First-Time-Right formulation for Phase I. Streamlining the transition to future clinical studies upon Phase I success, this innovative approach sets the foundation for optimized drug development and success beyond.
Home
Crystal Pharmatech's Mol2Med™ First-Time-Right approach guarantees a robust API form and a scalable manufacturing process, culminating in a First-Time-Right formulation for Phase I. Streamlining the transition to future clinical studies upon Phase I success, this innovative approach sets the foundation for optimized drug development and success beyond.
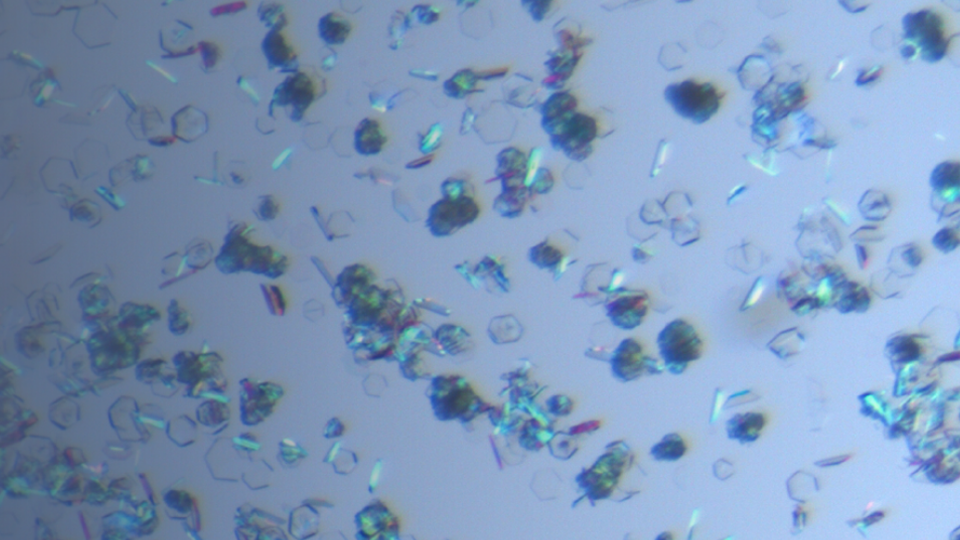 Solid Form Screening and Selection
Solid form is a general term that refers to both crystalline and amorphous materials. The solid form will impact active pharmaceutical ingredient (API) development properties such as solubility, dissolution rate, stability, hygroscopicity, and bioavailability.
Solid Form Screening and Selection
Solid form is a general term that refers to both crystalline and amorphous materials. The solid form will impact active pharmaceutical ingredient (API) development properties such as solubility, dissolution rate, stability, hygroscopicity, and bioavailability.
 Case Study 1: Indinavir - Early Salt Form Change
BackgroundIt has been reported that the solid form of active pharmaceutical ingredients (APIs) has significantly impacted quality and consistency of the final dosage form for drug development compound...
Case Study 1: Indinavir - Early Salt Form Change
BackgroundIt has been reported that the solid form of active pharmaceutical ingredients (APIs) has significantly impacted quality and consistency of the final dosage form for drug development compound...
 Biologics Characterization and CMC Analytics
CBS provides analytical and CMC services and solutions for the development, manufacturing and approval of new mAbs and therapeutical proteins and their biosimilars.
Biologics Characterization and CMC Analytics
CBS provides analytical and CMC services and solutions for the development, manufacturing and approval of new mAbs and therapeutical proteins and their biosimilars.
.webp) First-Time-Right 3-STEP Approach
Crystal Pharmatech are a specialized CRO/CDMO focused on Solid State Research, Pre-Formulation, Formulation Development and Manufacturing. Our strength is our focus and expertise in these specialties...
First-Time-Right 3-STEP Approach
Crystal Pharmatech are a specialized CRO/CDMO focused on Solid State Research, Pre-Formulation, Formulation Development and Manufacturing. Our strength is our focus and expertise in these specialties...
 Bioanalytical and Biomarker Services
With more than 100 years of combined industry experience of our leadership team and three GLP compliant laboratories, we are ready for all your current and emerging bioanalytical needs.
Bioanalytical and Biomarker Services
With more than 100 years of combined industry experience of our leadership team and three GLP compliant laboratories, we are ready for all your current and emerging bioanalytical needs.
 Holistic Polymorph Screening - Identify the Lead Crystal Form and Accelerate CMC Progress
From early developability to late-stage specifications—polymorph/salt-cocrystal strategy, SCXRD/MicroED solutions, solvent & pathway design, drug-product form control, ASD crystalline-form limits...
Holistic Polymorph Screening - Identify the Lead Crystal Form and Accelerate CMC Progress
From early developability to late-stage specifications—polymorph/salt-cocrystal strategy, SCXRD/MicroED solutions, solvent & pathway design, drug-product form control, ASD crystalline-form limits...
 DNA Synthesis
DNA Synthesis
 RNA Synthesis
RNA Synthesis
Subscribe to be the first to get the updates!