Olanzapine (Fig. 12), a Biopharmaceutics Drug Disposition Classification System (BDDCS) 2 drug (Benet et al. 2011) with poor solubility and high permeability, has been marketed towards treating schizophrenia. Olanzapine has been shown to exhibit a number of different crystalline forms including hydrates (Reutzel-Edens et al. 2003) and solvates (Cavallari et al. 2013). Form I has been deemed the most stable unsolvated form (Reutzel-Edens et al.2003). A variety of dosage forms have been developed to target different patient populations. These products have included Zyprexa® tablets (once a day oral tablets), Zyprexa Zydis® orally disintegrating tablets (that can be taken without water), and Zyprexa Intra Muscular® (rapid acting intramuscular injection). A combination capsule product with fluoxetine hydrochloride (HCl) (Symbyax®) was also launched when indications were expanded to include treatments of bipolar disorder and resistance depression in its marketing.
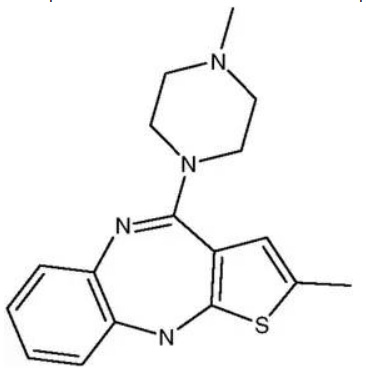
Fig. 12: Structure of olanzapine
A major issue with this patient group was compliance; as a result, a dosage form that lasted longer than once daily would have provided a significant benefit for the patients. To address this issue, researchers developed a long acting injection (LAI) using olanzapine pamoate monohydrate and sold as Zyprexa Relprevv® in 2010 (Chue and Chue 2012). The pamoate salt was shown to be poorly soluble in aqueous media, and micron sized crystals were suspended in a diluent containing carboxymethylcellulose sodium, mannitol, polysorbate 80, sodium hydroxide and/or hydrochloric acid for pH adjustment and water for injection (Zyprexa Relprevv Package Insert 2014). As a result, the salt slowly dissolved after injection into the muscle, resulting in an absorption of olanzapine systemically over a period of several weeks (Citrome 2009). The half-life of the pamoate salt became 30 days, in comparison to 33 h for an oral dose (Di Lorenzo and Brogli 2010). One injection has been noted to last three to four weeks, providing better efficacy and compliance for patients (Fig. 13) (Agency et al. 2015). The efficacy and tolerability profiles for the LAI were found to be the same as the oral formulation. The olanzapine Form II patent listed in the Orange Book (US 6960577) is set to expire in 2017. The olanzapine pamoate monohydrate patent listed in the Orange Book (US 6169084) has an expiry date of 2018, which has given the LAI dosage form a year of extra patent coverage.
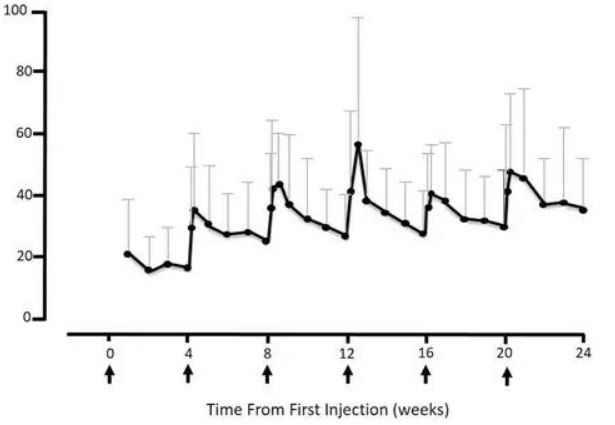
Fig. 13: Mean olanzapine plasma concentrations for the Multiple-Dose Group receiving 405 mg/4 weeks; arrows along x-axis indicate injections (adapted from reference (Agency et al. 2015))
This case study has illustrated the advantages of using novel solid forms for innovative drug products. The change in solid form to a crystalline pamoate salt resulted in a less soluble salt, which has previously not been desired by researchers. However, in this case, the less soluble salt exhibited all the properties needed for an improved sustained release formulation. Rather than an oral dosage form, an intramuscular injection was produced to capitalize on the lower solubility. The result became a dosage form with good efficacy and superior compliance. Additionally, patent coverage around the new salt has also extended coverage for a year after the olanzapine free base expires.
Subscribe to be the first to get the updates!