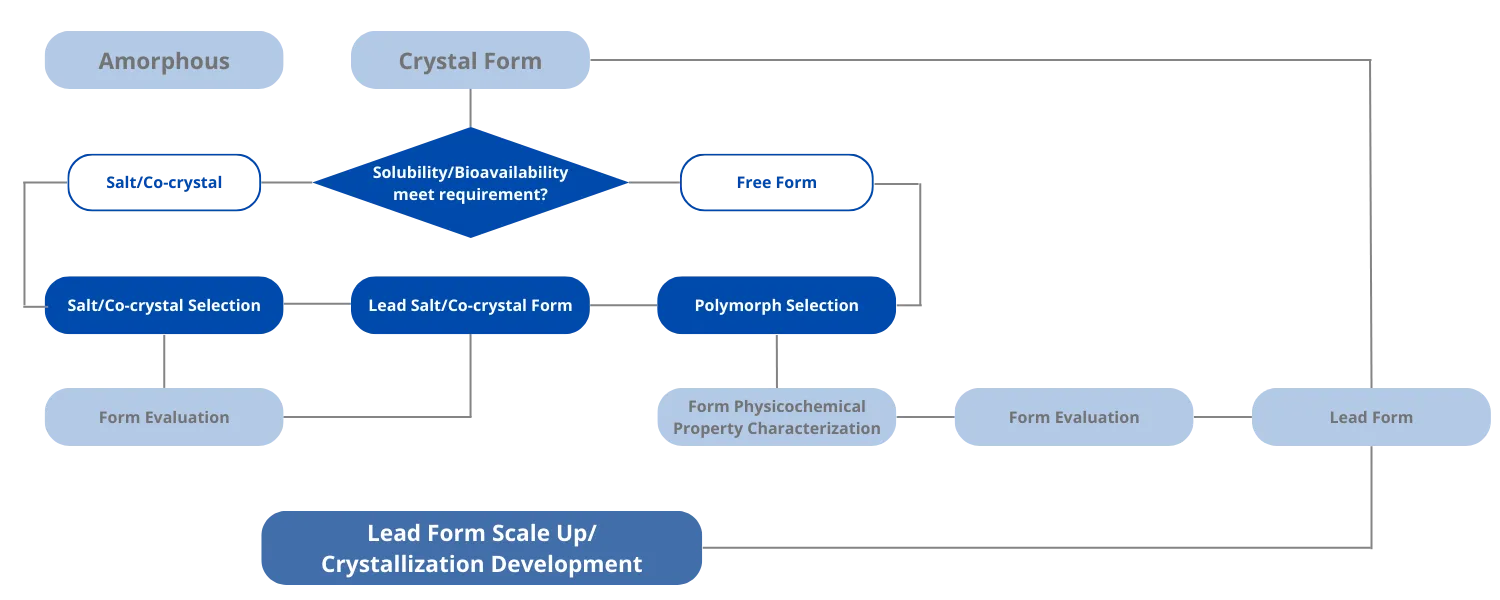
The selected solid form directly impacts solubility, stability, bioavailability, and manufacturability. Comprehensive screening is critical to ensuring product quality and clinical performance.
As a specialized CRO/CDMO in solid-state research, we provide integrated capabilities in physicochemical characterization to support polymorph, salt, and co-crystal selection.
Equipped to handle Highly Potent Compounds (OEB 4 & 5) and Controlled Substances.
Focused targeted manual screening for rapid assessment of key properties and initial form identification.
Integrated virtual (COSMO-RS) and automated experimental workflows to evaluate phase behavior and stability.
High-throughput screening for comprehensive form space evaluation to ensure robust control and IP protection.
Mapping the polymorphic landscape to identify the most thermodynamically stable form with optimal stability, hygroscopicity, and melting behavior.
Applied when improvements in solubility, stability, or bioavailability are required. Systematic evaluation enables identification for scalable development.
For non-ionizable or weakly ionizable compounds, co-crystallization provides an alternative strategy to modify physicochemical properties.
Have a question or need support with your project? Please complete the form, and our team will get back to you shortly.
Our capabilities span three specialized platforms:
Small Molecule
Crystal Bio Solutions
Crystal NAX
By providing your e-mail address, you agree to receive an e-mail response from Crystal Pharmatech to your inquiry. The information you submit will be governed by our Privacy Policy.
Subscribe to be the first to get the updates!