More than 70% of drug candidates are chiral. Crystallization provides a more economical and sustainable alternative to traditional chromatography for producing enantiopure compounds.
Separating desired enantiomers via diastereomeric salt formation and crystallization.
Surpassing the 50% yield limit by combining active racemization with targeted crystallization.
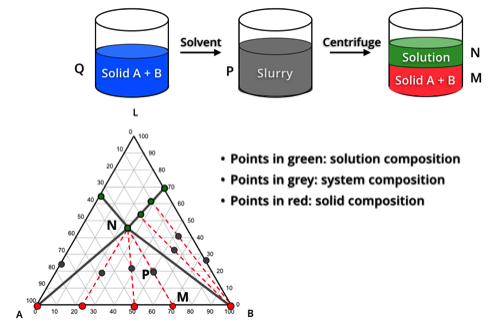
We do not rely on trial and error. Crystal Pharmatech utilizes Ternary Phase Diagrams as a powerful predictive tool to fundamentally understand system thermodynamics.
Have a question or need support with your project? Please complete the form, and our team will get back to you shortly.
Our capabilities span three specialized platforms:
Small Molecule
Crystal Bio Solutions
Crystal NAX
By providing your e-mail address, you agree to receive an e-mail response from Crystal Pharmatech to your inquiry. The information you submit will be governed by our Privacy Policy.
Subscribe to be the first to get the updates!